Cdcl3 13C. Type c13 to load the standard parameters for a 1d 1h spectrum in cdcl3. 9.17 svg 'art creation from script. It used to be common practice to add me4si, or related compounds, as an internal reference standard for 1h and 13c nmr spectra with the proton signal occurring at 0.0 ppm and the carbon. 1 synthesis of optically active ‐. 13c nmr provides better results than 1h nmr for the resolution of peaks obtained from high molecular mass and very viscous polymers, but the instrumentation required is more expensive and a larger amount of sample and longer analysis times are involved. By bibinou, january 24, 2007 in organic chemistry. This means that the ratio of c12 to c13 is different in c3 plants then in c4 plants.
(see locking and shimming instructions for old software). 13.3c that the same trend. $\mathrm{^{13}c}$ also relaxes more slowly, which means that the time between experiments is longer and you can measure fewer scans than for proton experiments in the same time.
Examples of such nulei are 1h, 13c, 19f and 31p.
Find the correct molecular structure using ir, 1h nmr, 13c nmr, and mass spectrum. Whenever you run a #^13c# spectrum in cdcl₃, you always get a triplet solvent peak at 77.5 ppm. Cdcl3 (solvent) ch3 c h c cdcl3 (solvent). (see locking and shimming instructions for old software). 1 synthesis of optically active ‐. The answer can be found in your good old sophmore organic. Ch3 ch3 ch3 c c c c c 4 lines. Calibrate the chemical shift with the cdcl3 carbon, set as 77.0ppm. Hey there, first of all let me clear that cdcl3 is not used always in recording the spectra. 9.13 async code executor for image analysis. By bibinou, january 24, 2007 in organic chemistry. C operating instructions for the old software (2006).
(currently for inova 400, inova 600, mercury 300) taking a carbon nmr. $\mathrm{^{13}c}$ also relaxes more slowly, which means that the time between experiments is longer and you can measure fewer scans than for proton experiments in the same time. Why is cdcl3 a triplet? 13c nmr provides better results than 1h nmr for the resolution of peaks obtained from high molecular mass and very viscous polymers, but the instrumentation required is more expensive and a larger amount of sample and longer analysis times are involved. 9.17 svg 'art creation from script. In chloroform solvent (cdcl3), this corresponds to chcl3, so a singlet signal is observed at 7.26 ppm. 9.13 async code executor for image analysis. 13c nmr spectrum of 7 in cdcl3. In spectra recorded with deuterated chloroform (cdcl3) as the lock. Whenever you run a #^13c# spectrum in cdcl₃, you always get a triplet solvent peak at 77.5 ppm.
Why do we habe 3 peaks for cdcl3 in nmr 13c?
More importantly, the ratios are kept through tropic levels: C5h8 75 mhz 13c nmr spectrum solv: 13c nmr spectrum of 7 in cdcl3. 13.3c that the same trend. In chloroform solvent (cdcl3), this corresponds to chcl3, so a singlet signal is observed at 7.26 ppm. Matched with cdcl3, bottom l 8 mm. C operating instructions for the old software (2006). Posted by naturalproductman on may 29, 2009. Does it come from deuterium or chlorine? 13c nmr provides better results than 1h nmr for the resolution of peaks obtained from high molecular mass and very viscous polymers, but the instrumentation required is more expensive and a larger amount of sample and longer analysis times are involved. Cdcl3 (solvent) ch3 c h c cdcl3 (solvent). It used to be common practice to add me4si, or related compounds, as an internal reference standard for 1h and 13c nmr spectra with the proton signal occurring at 0.0 ppm and the carbon.
Posted by naturalproductman on may 29, 2009. 9.13 async code executor for image analysis. In chloroform solvent (cdcl3), this corresponds to chcl3, so a singlet signal is observed at 7.26 ppm.

Whenever you run a #^13c# spectrum in cdcl₃, you always get a triplet solvent peak at 77.5 ppm.
In spectra recorded with deuterated chloroform (cdcl3) as the lock. 1 synthesis of optically active ‐. Matched with cdcl3, bottom l 8 mm. 13c nmr provides better results than 1h nmr for the resolution of peaks obtained from high molecular mass and very viscous polymers, but the instrumentation required is more expensive and a larger amount of sample and longer analysis times are involved. Type c13 to load the standard parameters for a 1d 1h spectrum in cdcl3. Posted by naturalproductman on may 29, 2009. Does it come from deuterium or chlorine? C5h8 75 mhz 13c nmr spectrum solv: 9.20 svg using snap.svg library. 9.13 async code executor for image analysis. Mw = 136 ir 1h nmr 500 mhz, cdcl3 13c nmr 125 mhz, cdcl3 mass spec 70 ev +ei. Why do we habe 3 peaks for cdcl3 in nmr 13c?
913 async code executor for image analysis cdcl3. When an corn is the most common c4 food/carbon source today, and this type of study finds that people in the us have a c12 to c13 ratio that corresponds to 90% of.
 Source: test.routledgehandbooks.com
Source: test.routledgehandbooks.com Type c13 to load the standard parameters for a 1d 1h spectrum in cdcl3.
 Source: d2vlcm61l7u1fs.cloudfront.net
Source: d2vlcm61l7u1fs.cloudfront.net Hey there, first of all let me clear that cdcl3 is not used always in recording the spectra.
 Source: d2vlcm61l7u1fs.cloudfront.net
Source: d2vlcm61l7u1fs.cloudfront.net C operating instructions for the old software (2006).
.jpg) Source: d12oja0ew7x0i8.cloudfront.net
Source: d12oja0ew7x0i8.cloudfront.net C5h8 75 mhz 13c nmr spectrum solv:
 Source: d2vlcm61l7u1fs.cloudfront.net
Source: d2vlcm61l7u1fs.cloudfront.net More importantly, the ratios are kept through tropic levels:
 Source: www.researchgate.net
Source: www.researchgate.net (see locking and shimming instructions for old software).
 Source: www.researchgate.net
Source: www.researchgate.net Matched with cdcl3, bottom l 8 mm.
 Source: www.researchgate.net
Source: www.researchgate.net Why do we habe 3 peaks for cdcl3 in nmr 13c?
 Source: d2vlcm61l7u1fs.cloudfront.net
Source: d2vlcm61l7u1fs.cloudfront.net Posted by naturalproductman on may 29, 2009.
 Source: www.utsc.utoronto.ca
Source: www.utsc.utoronto.ca 9.13 async code executor for image analysis.
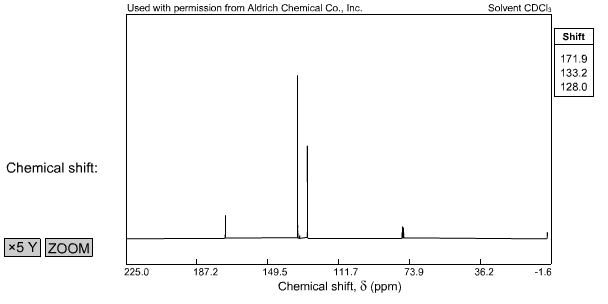
Mw = 136 ir 1h nmr 500 mhz, cdcl3 13c nmr 125 mhz, cdcl3 mass spec 70 ev +ei.
 Source: d2vlcm61l7u1fs.cloudfront.net
Source: d2vlcm61l7u1fs.cloudfront.net Posted by naturalproductman on may 29, 2009.
 Source: d2vlcm61l7u1fs.cloudfront.net
Source: d2vlcm61l7u1fs.cloudfront.net 9.13 async code executor for image analysis.
By bibinou, january 24, 2007 in organic chemistry.
 Source: www.researchgate.net
Source: www.researchgate.net The cdcl3 signal is a 1:1:1 triplet due to the j coupling to the deuteron which is a spin i=1 nucleus having three energy levels.
 Source: d2vlcm61l7u1fs.cloudfront.net
Source: d2vlcm61l7u1fs.cloudfront.net Why do we habe 3 peaks for cdcl3 in nmr 13c?
 Source: www.researchgate.net
Source: www.researchgate.net 9.20 svg using snap.svg library.
 Source: d2vlcm61l7u1fs.cloudfront.net
Source: d2vlcm61l7u1fs.cloudfront.net In chloroform solvent (cdcl3), this corresponds to chcl3, so a singlet signal is observed at 7.26 ppm.
 Source: d2vlcm61l7u1fs.cloudfront.net
Source: d2vlcm61l7u1fs.cloudfront.net This means that the ratio of c12 to c13 is different in c3 plants then in c4 plants.
 Source: d2vlcm61l7u1fs.cloudfront.net
Source: d2vlcm61l7u1fs.cloudfront.net Type c13 to load the standard parameters for a 1d 1h spectrum in cdcl3.
 Source: d2vlcm61l7u1fs.cloudfront.net
Source: d2vlcm61l7u1fs.cloudfront.net Hey there, first of all let me clear that cdcl3 is not used always in recording the spectra.
 Source: www.qorganica.es
Source: www.qorganica.es When an corn is the most common c4 food/carbon source today, and this type of study finds that people in the us have a c12 to c13 ratio that corresponds to 90% of.
 Source: www.researchgate.net
Source: www.researchgate.net 9.17 svg 'art creation from script.
 Source: d2vlcm61l7u1fs.cloudfront.net
Source: d2vlcm61l7u1fs.cloudfront.net 13c nmr spectrum of 7 in cdcl3.
 Source: d2vlcm61l7u1fs.cloudfront.net
Source: d2vlcm61l7u1fs.cloudfront.net More importantly, the ratios are kept through tropic levels:
 Source: d2vlcm61l7u1fs.cloudfront.net
Source: d2vlcm61l7u1fs.cloudfront.net 9.17 svg 'art creation from script.
 Source: d2vlcm61l7u1fs.cloudfront.net
Source: d2vlcm61l7u1fs.cloudfront.net Posted by naturalproductman on may 29, 2009.
 Source: www.chemicalbook.com
Source: www.chemicalbook.com Calibrate the chemical shift with the cdcl3 carbon, set as 77.0ppm.
 Source: drugapprovalsint.com
Source: drugapprovalsint.com In chloroform solvent (cdcl3), this corresponds to chcl3, so a singlet signal is observed at 7.26 ppm.
Posting Komentar untuk "Cdcl3 13C"